For the cosmic chemistry project my group and I were asked to create a model of our choosing, that exemplified 1 to 3 learning target aspects of cosmic chemistry. The learning targets we had to choose from were as follows:
o Develop and use a model to explain changes in the composition of the nucleus and energy
released during the processes of fission, fusion and radioactive decay.
o Illustrate the energy transfer mechanisms that allow energy from nuclear fusion in the sun’s
core to reach Earth.
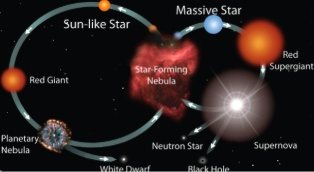
o Communicate scientific ideas about the way stars over their life cycle, produce elements.
We decided to focus on the third target listed involving star life cycle and how stars transform in their cycles. We centered our research on making a model to determine what classification any star could have once we research it. Some of the possible models we were given as examples on how to start were as followed, a computer simulation, online dating profile, song, story, picture, or a music video. We took a simpler approach.
Background-Before starting our final model, we did a lot of background research on stars and the universe. The first background assignment that we did was researching the big bang and how the elements in our universe were formed. We also made pre research models to see how we improved over time. Some of the facts that we found included:
o Develop and use a model to explain changes in the composition of the nucleus and energy
released during the processes of fission, fusion and radioactive decay.
o Illustrate the energy transfer mechanisms that allow energy from nuclear fusion in the sun’s
core to reach Earth.
o Communicate scientific ideas about the way stars over their life cycle, produce elements.
We decided to focus on the third target listed involving star life cycle and how stars transform in their cycles. We centered our research on making a model to determine what classification any star could have once we research it. Some of the possible models we were given as examples on how to start were as followed, a computer simulation, online dating profile, song, story, picture, or a music video. We took a simpler approach.
Background-Before starting our final model, we did a lot of background research on stars and the universe. The first background assignment that we did was researching the big bang and how the elements in our universe were formed. We also made pre research models to see how we improved over time. Some of the facts that we found included:
- Universe is 13.7 billion years old, Started with a Big Bang, 4.5 billion years ago: solar system formed, Baryonic matter: physical matter formed from baryonic particles (5%), Baryons type of hadrons (baryons are composite particles of quarks, such as protons, neutrons)
- Elementary Particles:
- Quarks: up, charm, top, bottom, down, strang
- Leptons: electron, muon, tau, electron neutrino, muon neutrino, tau neutrino
- Gauge Bosons: photon, W boson, Z boson, gluon
- Scalar Boson: Higgs
Dark Matter: cannot be observed using normal methods; gravitational effects on universe; rotational velocity of galaxies same at all places (27%)
Dark Energy: causes the expansion of the universe at a faster pace (68%)
Epochs of time - 4 Fundamental Forces:
- Gravity - mass attracts
- Electromagnetism - electrical interactions, magnetism
- The Weak Force - causes beta decay
- The Strong Force - keeps nucleons bound together
Our presentation is very strong in explaining the differences and characteristics of stars at different points in their life. This would be useful in identifying a star based on its observable characteristics,
https://docs.google.com/presentation/d/1Mt1X5H1rPMWqSjaGywrqtnVqzCPTFcC6rcee73EiGRE/edit?usp=sharing
https://docs.google.com/presentation/d/1Mt1X5H1rPMWqSjaGywrqtnVqzCPTFcC6rcee73EiGRE/edit?usp=sharing
My pre research knowledge model 1 and model 2.
So much Content-
I googled these
Law of Conservation of Mass: Matter is not created nor destroyed in any chemical or physical change
Law of definite proportions: a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the sample or source of the compound
Law of Multiple Proportions: if two or more different compounds are composed of the same two elements, then the ratio of the masses of the second element combined with a certain mass of the first element is always a ratio of small whole numbers
John Dalton's Atomic Theory
1) all matter is composed of atoms
2) all atoms of the same atom are identical
3) when compounds from, atoms combine in simple whole number ratios by mass(LODP)
4) atoms are either rearranged, combined or put together
5) atoms cannot be divided, created or destroyed
Joseph John Thomson: discovered the electron, "pudding" version of atom
Robert Millikan: used the oil-drop apparatus to determine the charge of an electron
Ernest Rutherford: Conducted the famous Gold foil experiment, discovered the nucleus of atoms
Subatomic Particles: a particle that is smaller than an atom
Fission: the splitting of an atomic nucleus to release energy
Fusion: creation of energy by joining the nuclei of two hydrogen atoms to form helium
Radioactive Decay: the spontaneous breakdown of an atomic nucleus resulting in the release of energy and matter from the nucleus
Alpha Decay: radioactive decay by emission of an alpha particle- He-4 (two protons and two neutrons)atomic number
Half-Life: length of time required for half of the radioactive atoms in a sample to decay. This relates to our project as stars have different half lives which allow them to decay at fast or slower paced rates.
Nuclear Transmutation: can be induced by accelerating a particle to collide it with the nuclide
Radiation: the emission of energy as electromagnetic waves or as moving subatomic particles, especially high-energy particles that cause ionization
Nucleosynthesis: the process that creates new atomic nuclei from pre-existing nucleons, primarily protons and neutrons (atoms slam together to form reactions)
Alpha Particle: a helium nucleus emitted by some radioactive substances, originally regarded as a ray
Beta Particle: a fast-moving electron emitted by radioactive decay of substances
Gamma Emission: when an excited nucleus gives off a ray in the gamma part of the spectrum; has no mass and no charge
Positron Emission: a radioactive decay process that involves the emission of a positron, a particle that has the same mass but opposite charge to an electron
Electron Capture: an inner orbital electron is captured by the nucleus of its own atom
Photon: a particle of electromagnetic radiation with no mass that carries a quantum of energy
Excited State: a state in which an atom has a higher potential energy than it has in its ground state
Ground State: the lowest energy state of an atom
Ion: an atom or group of atoms that has a positive or negative charge- changed number of electrons
Isotope: atoms of the same element that have different numbers of neutrons
Supernova: when the outward forces of pressure win out over the inward force of gravity causing a massive luminous explosion.
Black Hole: when the inward force of gravity wins out over the outward pressure, inversely from a supernova a black hole collapses and doesn't allow anything (light, particles...) to escape from it.
end of content except for content found throughout our presentation
---------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
Reflection-
Two things that I did well in the group part of this project were creative thinking and time management. The first skill that I used in this project was creative thinking an example of how is when my group and I were deciding on how to compose the models of types of stars We knew that we wanted to have a realistic model that can bring an easy understanding of star life cycles to all groups and ages. In our brainstorming we worked to narrow down how we would make the different stars and show them and we settled with a website that is a 3D “modeling” platform that allowed to create unique simplistic designs of stars. The second skill that I utilized in this project was time management. A good example of when we used this is how my group divided and conquered with the different models we had 3 people and 10 or so models so we just assigned a few to each other and got them done. This skill was very useful for us and It will continue to let us thrive in the future.
Two skills I need to work on for the next project are communication and a thought provoking demeanor. The first skill I will need to work on for my next group would be my communication. During the weeks that we had I feel like we didn't communicate back and forth about our slide show we’d have to present. An example is when presenting some of us didn't know the vocab words we were teaching the class about. In the future I will make sure that my group and I communicate and talk these easy to fix issues before we get to the point where we cannot solve the problem. The second skill that I still need to work on is thought provoking demeanor. What I mean by this is there should’ve been many more things our group shared with the class about and had we thought about those things and had slides for said things then we could’ve made an even more educational experience. This is what I call a conscientious learning movement. We also could’ve made our bibliography much okay-er.
All in all, this was a really fun project to work on! I was able to learn more about our universe and how it came to be, how elements were formed, and about stars life cycles. I enjoyed learning more about the origins of these concepts and also working on developing my understanding through a hands on star life cycle determiner.
I googled these
Law of Conservation of Mass: Matter is not created nor destroyed in any chemical or physical change
Law of definite proportions: a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the sample or source of the compound
Law of Multiple Proportions: if two or more different compounds are composed of the same two elements, then the ratio of the masses of the second element combined with a certain mass of the first element is always a ratio of small whole numbers
John Dalton's Atomic Theory
1) all matter is composed of atoms
2) all atoms of the same atom are identical
3) when compounds from, atoms combine in simple whole number ratios by mass(LODP)
4) atoms are either rearranged, combined or put together
5) atoms cannot be divided, created or destroyed
Joseph John Thomson: discovered the electron, "pudding" version of atom
Robert Millikan: used the oil-drop apparatus to determine the charge of an electron
Ernest Rutherford: Conducted the famous Gold foil experiment, discovered the nucleus of atoms
Subatomic Particles: a particle that is smaller than an atom
Fission: the splitting of an atomic nucleus to release energy
Fusion: creation of energy by joining the nuclei of two hydrogen atoms to form helium
Radioactive Decay: the spontaneous breakdown of an atomic nucleus resulting in the release of energy and matter from the nucleus
Alpha Decay: radioactive decay by emission of an alpha particle- He-4 (two protons and two neutrons)atomic number
Half-Life: length of time required for half of the radioactive atoms in a sample to decay. This relates to our project as stars have different half lives which allow them to decay at fast or slower paced rates.
Nuclear Transmutation: can be induced by accelerating a particle to collide it with the nuclide
Radiation: the emission of energy as electromagnetic waves or as moving subatomic particles, especially high-energy particles that cause ionization
Nucleosynthesis: the process that creates new atomic nuclei from pre-existing nucleons, primarily protons and neutrons (atoms slam together to form reactions)
Alpha Particle: a helium nucleus emitted by some radioactive substances, originally regarded as a ray
Beta Particle: a fast-moving electron emitted by radioactive decay of substances
Gamma Emission: when an excited nucleus gives off a ray in the gamma part of the spectrum; has no mass and no charge
Positron Emission: a radioactive decay process that involves the emission of a positron, a particle that has the same mass but opposite charge to an electron
Electron Capture: an inner orbital electron is captured by the nucleus of its own atom
Photon: a particle of electromagnetic radiation with no mass that carries a quantum of energy
Excited State: a state in which an atom has a higher potential energy than it has in its ground state
Ground State: the lowest energy state of an atom
Ion: an atom or group of atoms that has a positive or negative charge- changed number of electrons
Isotope: atoms of the same element that have different numbers of neutrons
Supernova: when the outward forces of pressure win out over the inward force of gravity causing a massive luminous explosion.
Black Hole: when the inward force of gravity wins out over the outward pressure, inversely from a supernova a black hole collapses and doesn't allow anything (light, particles...) to escape from it.
end of content except for content found throughout our presentation
---------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------
Reflection-
Two things that I did well in the group part of this project were creative thinking and time management. The first skill that I used in this project was creative thinking an example of how is when my group and I were deciding on how to compose the models of types of stars We knew that we wanted to have a realistic model that can bring an easy understanding of star life cycles to all groups and ages. In our brainstorming we worked to narrow down how we would make the different stars and show them and we settled with a website that is a 3D “modeling” platform that allowed to create unique simplistic designs of stars. The second skill that I utilized in this project was time management. A good example of when we used this is how my group divided and conquered with the different models we had 3 people and 10 or so models so we just assigned a few to each other and got them done. This skill was very useful for us and It will continue to let us thrive in the future.
Two skills I need to work on for the next project are communication and a thought provoking demeanor. The first skill I will need to work on for my next group would be my communication. During the weeks that we had I feel like we didn't communicate back and forth about our slide show we’d have to present. An example is when presenting some of us didn't know the vocab words we were teaching the class about. In the future I will make sure that my group and I communicate and talk these easy to fix issues before we get to the point where we cannot solve the problem. The second skill that I still need to work on is thought provoking demeanor. What I mean by this is there should’ve been many more things our group shared with the class about and had we thought about those things and had slides for said things then we could’ve made an even more educational experience. This is what I call a conscientious learning movement. We also could’ve made our bibliography much okay-er.
All in all, this was a really fun project to work on! I was able to learn more about our universe and how it came to be, how elements were formed, and about stars life cycles. I enjoyed learning more about the origins of these concepts and also working on developing my understanding through a hands on star life cycle determiner.